
2022 End of Year Message from the President of the Penta Foundation, Carlo Giaquinto
The impact of variant and vaccination on SARS-CoV-2 symptomatology; three prospective household cohorts

Get to know the UNIVERSAL project: Assessing the long-term safety of ART drugs

PentaTr@ining: ‘Fungal Infections in Paediatrics’ online course wraps up!
Implementation of infection prevention and control for hospitalised neonates: A narrative review

WHO publishes the AWaRe (Access, Watch, Reserve) Antibiotic Book

Experts call for urgent action to develop antibiotics for newborns

🇬🇧 Experts call for urgent action to develop antibiotics for newborns

‘PentaTr@ining: Viral Hepatitis in Children’ – online course launches!

World AIDS Day 2022: Is HIV cure attainable in our lifetime?
Antibiotics needed to treat multidrug-resistant infections in neonates

World Antimicrobial Awareness Week: tackling antimicrobial resistance in children and neonates
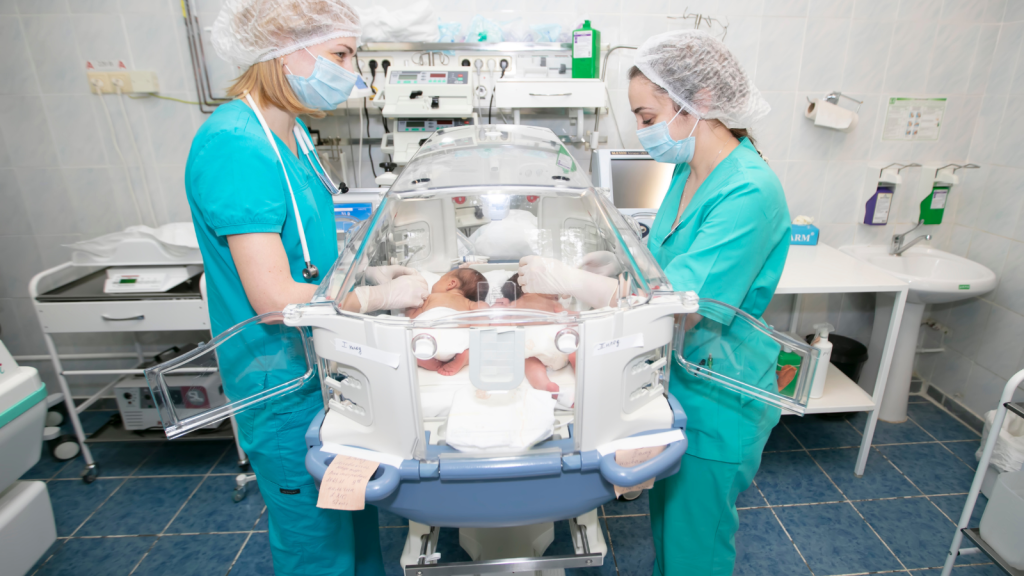
NeoIPC Clinical Practice Network kicks off its activities

A glimpse into a Penta Youth Trial Boards Meeting

EPIICAL General Assembly meeting to reaffirm mission to achieve HIV remission in children

Join our next Penta Aperitivo on 10 November 2022!
